An outstanding source of trial data lacking from publications: IQWiG assessment reports. The NIAGARA trial example.
Reports from HTA bodies may contain key data absent from trial publications and constitute a unique source for trial appraisal.
An underappreciated source of trial data, which may be lacking in publications, are public reports from various Health Technology Assessments (HTA) and regulators. Among the numerous and valuable sources, the German HTA body, The Institute for Quality and Efficiency in Healthcare (IQWiG), is one agency I want to highlight, as they are requiring key aspects of the data to be submitted to their agency and, as a result, you can find those data in their public reports.
Where are subsequent therapy data from NIAGARA?
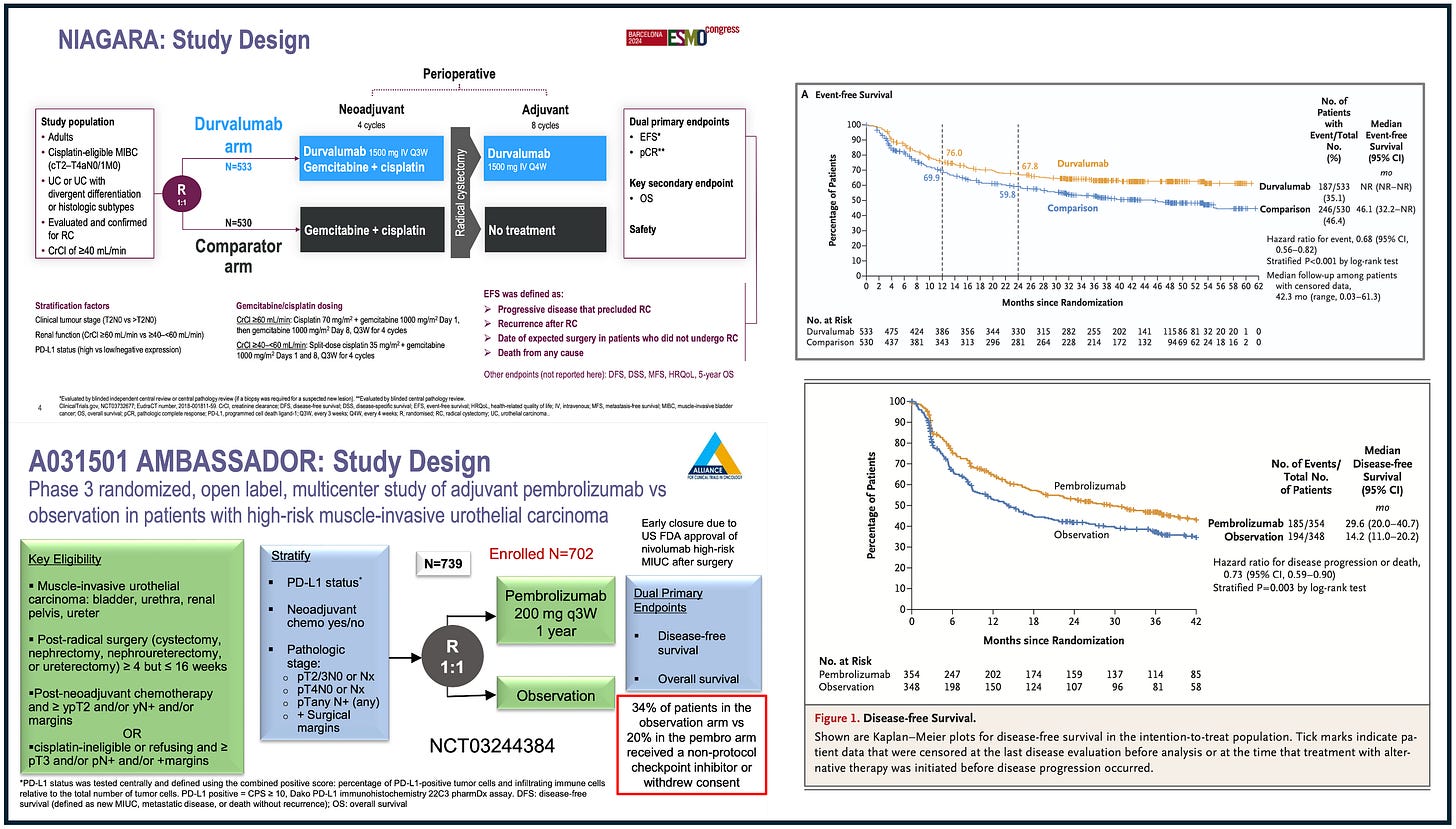
In a previous video, I made a side-by-side comparison between NIAGARA and AMBASSADOR trials (video available here). Both trials were run in the peri-operative setting of bladder cancer (NIAGARA is neo-adjuvant followed by adjuvant, AMBASSADOR is purely adjuvant). Both trials demonstrated an EFS/DFS gain in the experimental arm. Trials’ design (left) and their corresponding EFS/DFS results (right) are shown below:
However, only NIAGARA demonstrated an overall survival gain. The side-by-side comparison was primarily aimed at highlighting that, in global trials like NIAGARA, a survival gain may be driven by the limited access to optimal care for patients who recur.
Such limited access is explained when some enrolling countries are simply lacking access to the best care outside the trial. Conversely, in trials like AMBASSADOR, that was exclusively run in the US, optimal access to post-protocol care, which is desirable!, may have explained the absence of survival gain.
In a paper published in the European Journal of Cancer (full paper here), we posit that such global trials are rewarded in terms of guideline recommendations and approvals.
Here is the side-by-side comparison showing how NIAGARA was “rewarded” as compared to AMBASSADOR.
The subsequent therapy data from NIAGARA are now available in the IQWiG assessment report!
Even though the discussion with the NIAGARA trialists during the first presentation in September 2024 suggested that post-protocol access to immune checkpoint inhibitors would likely be low, we were awaiting for those data until very recently.
They are still, to my knowledge, unpublished.
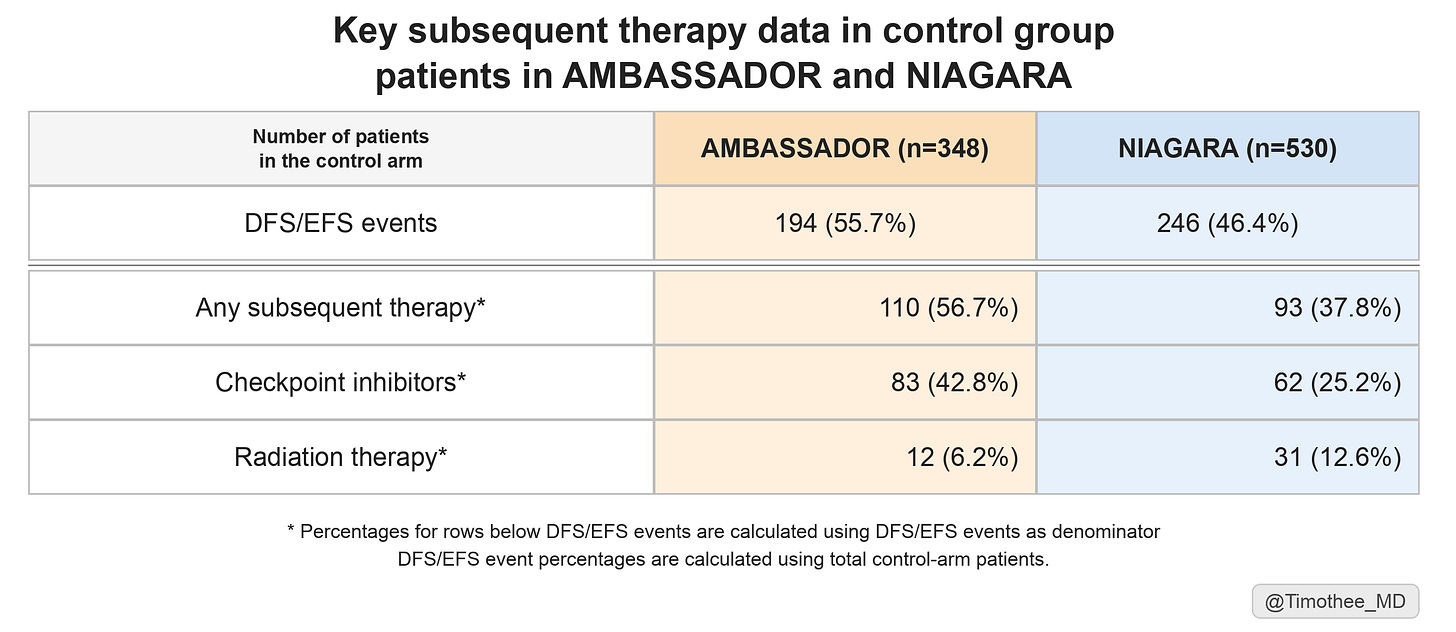
However, NIAGARA was recently evaluated by IQWiG. In their assessment, some key subsequent therapy data are now reported. Based on those, we can also compare them to AMBASSADOR (see table below).
A common, yet misleading, argument is to use real-world data (often involving frailer patients) to explain the low rates of post-protocol care in trials, when the underlying cause is limited global access. A more appropriate comparison is with trials such as AMBASSADOR, which are highly valuable to illustrate what can be expected as optimal subsequent therapy in a clinical trial setting.
Here, we can derive two conclusions for patients in the control group of NIAGARA:
confirmation of a very limited access to checkpoint inhibitors, received by only 25% of patients with an EFS event.
a limited access to any subsequent therapy, 38% of patients with an EFS event in NIAGARA, significantly lower than 57% of patients with a DFS event in AMBASSADOR.
Both may have biased the trial toward an overall survival gain in NIAGARA.
Why we should follow IQWiG and other HTA bodies reports.
It should be noted that other HTA bodies may also be providing key data. In the case of NIAGARA, the CDA-AMC Canadian agency also reported subsequent therapy in their report (available here, Table 8).
A specificity is that IQWiG is also systematically requiring the first subsequent treatment as well. Subsequent therapy data lacking granularity can confuse their interpretation (we have written about this together with Vinay Prasad, see here)
The sponsors, when submitting a dossier to IQWiG, must specifically provide :
“details regarding subsequent treatments administered after discontinuation of the study medication (for oncology-related issues, please also provide separate details regarding the first subsequent treatment).”
They are also requiring, systematically, the numbers of censored patients:
“ In survival analyses, the Kaplan-Meier curve should be plotted, including information on patients at risk and the proportion of censored patients over time (at multiple time points). A separate Kaplan-Meier curve should be plotted for each endpoint for which such an analysis is performed.”
IQWiG is doing an outstanding work. Their reports, which are publicly available, are usually translated into English a couple of months after publication. English reports are available on the IQWiG website, switching to english language, and searching for the relevant drugs. Definitely worth following closely!






Agree. IQWiG has one of the best reports… very transparent. No redactions like you see with NICE reports.
Hey — I came across your writing and really liked how you think.
I’m exploring something similar from a different angle — writing about human behavior through a system design lens (like debugging internal patterns).
Just started publishing on Substack. If you ever get a moment to read, I’d genuinely value your perspective.
Also happy to support your work — feels like there’s an interesting overlap here.