RASolute 302 (daraxonrasib) in metastatic pancreatic cancer: a revolution?
A pharmacological breakthrough might not always revolutionize patients' outcomes.
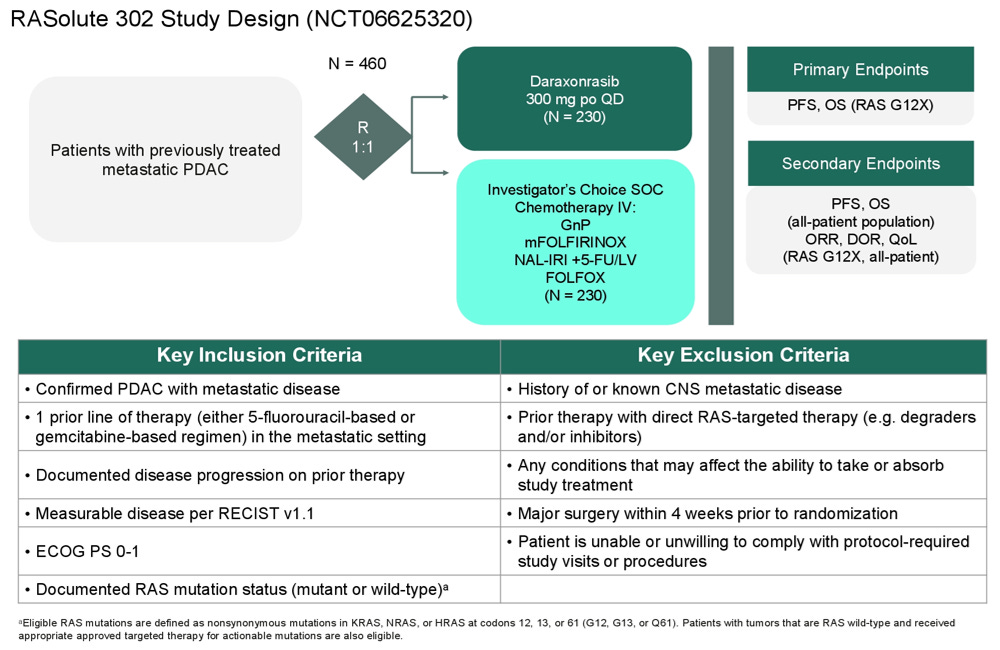
On April 13, 2026, the company Revolution Medicines announced the positive results of the phase 3 trial RASolute 302 in pancreatic cancer via a press-release (here). The trial was a multicenter global phase 3 trial, conducted in the US, France, Germany, Italy, Japan, Puerto Rico and Spain (see here). It included patients with metastatic pancreatic cancer who had previously received one prior line, in the metastatic setting, of therapy with 5-fluorouracil (5-FU) based or gemcitabine-based regimen.
In RASolute 302, patients either received daraxonrasib, an oral RAS(ON) multi-selective, non-covalent inhibitor; or chemotherapy.
More than 90% of patients with metastatic pancreatic cancer have a RAS mutation with about 85% carrying a KRAS G12 mutation. Contrary to other KRAS inhibitors, daraxonrasib inhibits the active RAS(ON) form. Even RAS-wild-type tumors may be dependent on RAS activation, which is the rationale for investigating daraxonrasib beyond RAS-mutant populations.
The press-release says: “In the overall (intent-to-treat) study population, daraxonrasib demonstrated a median OS of 13.2 months versus 6.7 months for chemotherapy, with a hazard ratio of 0.40 (p < 0.0001). Daraxonrasib was generally well tolerated, with a manageable safety profile and with no new safety signals.”
The results will be presented in a few weeks, during ASCO2026 plenary session, and a simultaneous publication in the New England Journal of Medicine would not be surprising.
Most doctors, scientists, and academics agree that we should not practice medicine by press release. However, following the RASolute 302 press release, social media and the press have echoed the results, mostly within a one-sided framing and an abundance of superlatives, presenting daraxonrasib as a “miracle” or a “revolution.”
While doubling median survival in patients with such a poor prognosis is highly unusual, caution is warranted when interpreting any results. Here are some initial thoughts, and what I think we should be looking for during ASCO.
Medicine by press-release? Why this is problematic.
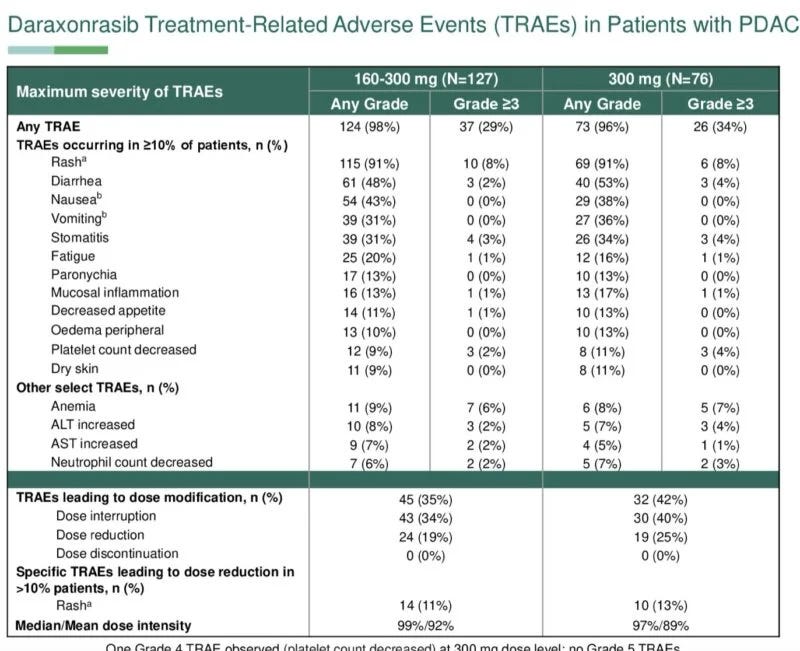
If we look at the toxicity data for daraxonrasib given as monotherapy, as in RASolute 302, data from patients with advanced pancreatic cancer treated in the phase 1/1b study were reported in a previous Revolution Medicines press-release issued in December 2024 (screenshot).
A quick read gives the false impression that there is minimal grade 3 or higher treatment-related toxicity.
This wording indicates that no individual adverse event exceeded 10% of patients. What is missing from the press-release is that, when taken together, approximately 34% of patients in the 300 mg cohort experienced grade 3 or higher treatment-related adverse events (see below).
In other words, this does not mean low toxicity, but rather distributed toxicity.
Toxicity data will be much awaited from the ASCO 2026 presentation. We can already get a glimpse of how “nasty” the drug can be. Here, I’m quoting Senator Ben Sasse, who has publicly shared his personal story after being diagnosed with advanced pancreatic cancer and enrolling in RASolute 302.
Ben Sasse : “I take it orally, but it’s a nasty drug. It causes crazy stuff like my body can’t grow skin and so I bleed all out of a whole bunch of parts of me that shouldn’t be bleeding.”

Former Senator Ben Sasse has given numerous interviews describing what he is going through as a patient with advanced pancreatic cancer. He has explicitly mentioned pain reduction and significant tumor shrinkage, and noted that “this experience has seemingly extended both quantity and quality of life” (here). At the same time, his public appearances and recounting also highlight the significant toxicities associated with the treatment.
So when we come back to the most recent press release from Revolution Medicine: “Daraxonrasib was generally well tolerated, with a manageable safety profile and with no new safety signals.” I wonder if patients, including Ben Sasse, would find that this wording is accurately reflecting his and other patients’ general experience.
Medicine by press-release? If we have no choice, let’s do it!
Let’s gather data from the press-release and from clinicaltrials.gov, and let’s see what we can make of it:
the trial included 500 patients, unselected for specific RAS mutations
primary endpoints = PFS and OS in RAS-G12-mutant patients
secondary endpoints = OS and PFS in all patients
“Trial met all primary and key secondary endpoints, including progression-free survival and overall survival”
“In the overall (intent-to-treat) study population, daraxonrasib demonstrated a median OS of 13.2 months versus 6.7 months for chemotherapy, with a hazard ratio of 0.40 (p < 0.0001).”
Study start (first patient): October 16, 2024; Enrollment (500 patients) completed: at least in December 2025; Total accrual duration: maximum 14 months; Press release 13 April 2026 (data-cut off likely in March 2026); Maximum follow-up duration about 17 months
The headline OS result is based on a secondary endpoint in the broadest patient population, while the primary endpoints, PFS and OS in the KRAS G12-mutant population, remain unreported
Why the OS estimates in the entire population could be overestimated.
Several factors may have contributed to an inflation of the reported results.
Power is usually calculated for the primary endpoint(s). As such, OS in the ITT population may be insufficiently powered. Additionally, hierarchical testing can reduce the available alpha, effectively reducing power in the ITT analysis.
Nested versus adjacent subgroups analyses. As the primary endpoints are in the RAS G12-mutant population, one key question for the overall population is: are non–G12 RAS-mutant patients truly deriving benefit, or is the observed effect in the “overall population” mainly driven by the G12-mutant population?
Early stopping rules carry a risk of inflating effect estimates, as shown in empirical work. Were such rules used in RASolute 302?
Efficacy–effectiveness gap? Patients in clinical trials often do not reflect real-world patients, who typically derive less benefit and may experience more toxicity. This is referred to as the efficacy–effectiveness gap. Even if the reported survival benefit is considered reliable, how it will translate into real-world practice remains an open question.
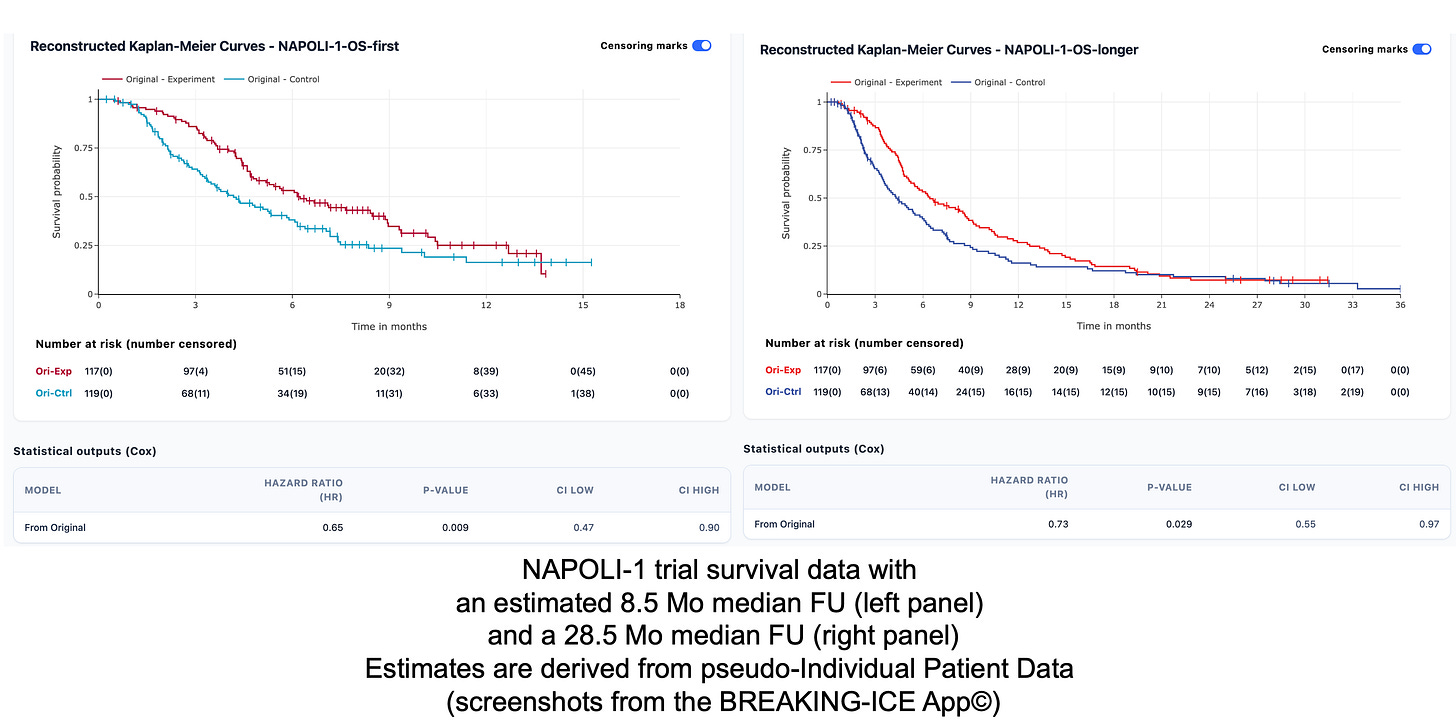
Longer follow-up matters: the NAPOLI-1 example.
Will longer follow-up data will ultimately be made available. The question is real, as the results are presented as being “primary and final”.
To illustrate the effect of longer follow-up, here is the NAPOLI-1 trial as an example, where we have two data points from the 2016 and 2019 reports. NAPOLI-1 was conducted in a setting comparable to RASolute 302. As shown in the estimations below, with longer follow-up, an early separation tends to diminish, with a less pronounced statistical gain.
This illustrates how early apparent benefit may attenuate with longer follow-up.
Key questions for ASCO and the full publication
Statistical details: early stopping rules if specified, alpha spending for each endpoint (particularly the ITT secondary endpoints), number of interim analyses planned, etc.
What is the median follow-up and OS maturity, what about censoring?
The shape of Kaplan–Meier curves: early separation ? curve crossing ? any tail?
ITT vs primary endpoints population: how much of the OS benefit in the entire ITT population may have been driven by the prespecified RAS-G12 population
Is the objective response rate (ORR) maintained? In the phase 1/1b study, the confirmed ORR was 35% in the RAS G12 population and 29% in the overall population. Classically, ORR tends to decrease from early-phase to later-stage studies. As a reminder, sotorasib (a KRAS G12C inhibitor) had an ORR of 37.1% in the early-phase CodeBreaK 100 trial, which later decreased to 28.1% in CodeBreaK 200.
Toxicity data!
Quality of life data: if presented: how and when they were measured, compliance rate, timing of measurements, etc.
Long-term efficacy? Drugs facing cancer biology
In 2011, George Sledge highlighted that the genomic era could bring both promise and chaos. He distinguished “stupid” cancers, like those driven by a single driver mutation, such as chronic myeloid leukemia (CML). In these settings, inhibitors can be transformative: in CML, life expectancy became comparable to the general population after the introduction of imatinib.
Yet, most solid tumors are different. They rarely rely on a single driver, and instead harbor co-mutations and multiple clones. In advanced disease, resistance at some point is the rule rather than the exception. Although daraxonrasib does not target a single mutation but rather RAS across multiple contexts, advanced pancreatic cancer remains a biologically “smart” cancer.
To which extent a broad RAS(ON) inhibition can temporarily suppress the genomic and adaptive complexity that usually drives resistance in pancreatic cancer is unclear.
My (temporary) conclusion.
Even though I’m awaiting for the data, I see daraxonrasib as a pharmacological breakthrough, however associated with significant toxicities and inherent biological hurdles when it comes to long-term efficacy.
On social media, discussions have at times become emotional, with any critical appraisal of a trial being perceived as a lack of empathy, which is unfortunate. We should not forget that financial stakes are high. Personal stories should not replace careful interpretation of the data.
In oncology, we often say a drug should help patients live longer and/or better. In the case of daraxonrasib, even with a survival benefit, the toxicity profile and the uncertainty around long-term benefit may make the choice not as straightforward for a fraction of patients.
Whether a treatment that delays, but does not prevent, death, while adding significant toxicity, constitutes a “revolution” is a question best answered by patients, not by Key Opinion Leaders (KOLs) or companies.