The Imbalance of NCCN Guidelines: Quick to Implement, Slow to Let Go
It’s my great pleasure to welcome Christopher Rios to The Oncology Shot. Chris is a brilliant medical student at UCSF, with whom we have worked on several projects within the VKPrasadLab. Congrats on this excellent paper, Chris (et al.), the stage is yours! Timothée
In our new paper, published in the Journal of Cancer Policy and openly accessible here, we analyze how and how quickly the National Comprehensive Cancer Network (NCCN) updates its guidelines in response to Food and Drug Administration (FDA) approvals and withdrawals over time.
The NCCN produces some of the most influential and referenced oncology guidelines worldwide. These guidelines strongly influence hematologists’ and oncologists’ clinical decision-making, and in the United States, when the NCCN recommends a drug, it regularly becomes covered by Medicare, Medicaid, and private insurers.
Given this influence, we were curious about how quickly NCCN updates its recommendations following regulatory decisions, as these updates likely affect patients, clinicians, and payors.
The Question: Do Guidelines Keep Pace with Regulatory Reality?
We examined how quickly the NCCN updates its guidelines after decisions from the U.S. Food and Drug Administration. Specifically, we looked at 83 FDA cancer drug approvals and 8 FDA withdrawals between October 2022 and July 2024. We also subdivided those FDA approvals into traditional regular approvals and accelerated approvals.
Our main findings:
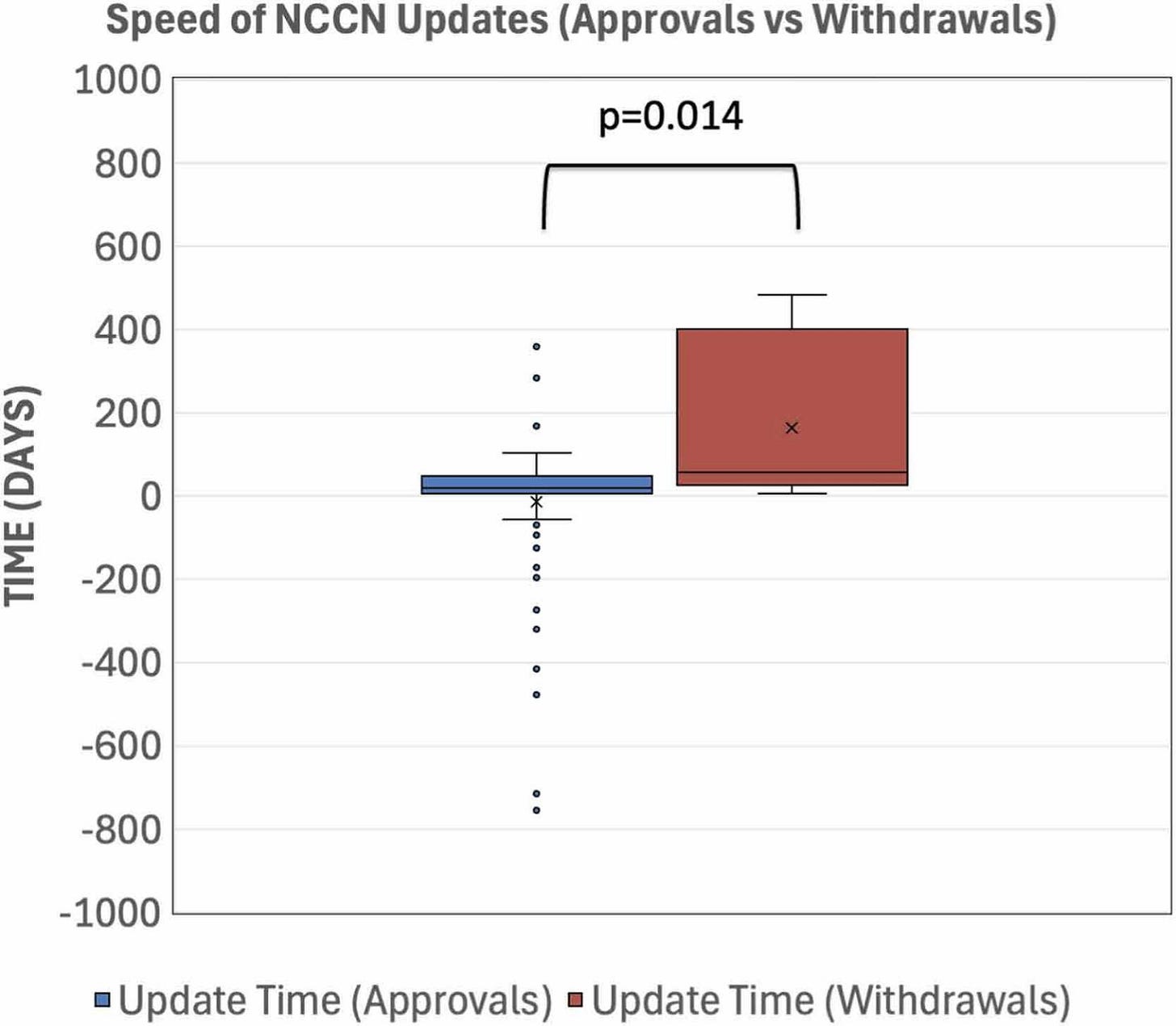
The NCCN incorporated FDA approvals very quickly, with a median of 19 days post-approval. In contrast, the NCCN updated withdrawn indications significantly slower, with a median of 57 days post-approval.
The NCCN removed only 3 of 8 withdrawn indications from its guidelines. The majority (5 of 8) remained recommended in some form, and four of these were recommended with either uniform or majority NCCN expert panel consensus (Category 2A and 2B recommendations).

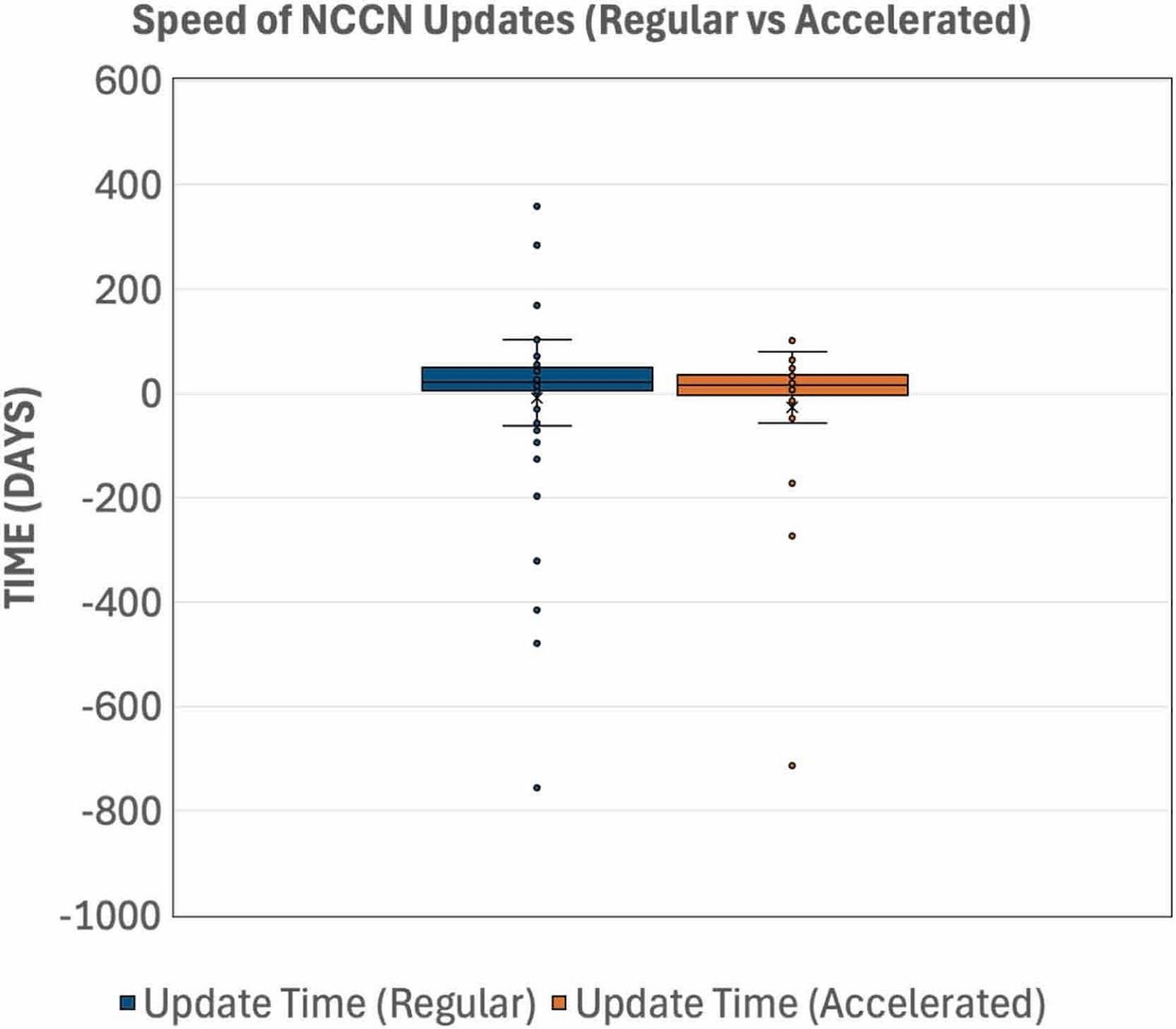
We found no significant differences in the time to NCCN update between regular and accelerated approval statuses, with median times of 20 days and 16 days, respectively.
Further insights into how NCCN panels make decisions
We also examined the NCCN’s recommendations for prescribing these therapies. We found that only one FDA-approved treatment (1.2%) included in the NCCN guidelines lacked uniform expert consensus (>85%) as a category 2B recommendation, which still comes with majority consensus (>50%).
This finding suggests that NCCN experts largely support the FDA’s approval decisions, in contrast to their attitude toward withdrawals as highlighted above.
As we point out in our discussion, this tendency toward universal consensus raises concerns, especially since 38% of all FDA approvals and nearly 80% of accelerated approvals come from single-arm, uncontrolled studies (see here).
Additionally, we found that in 22% of cases, the NCCN made additions or modifications to recommendations before receiving FDA approval. This does not imply that the NCCN is bypassing the FDA. Both the NCCN and the FDA respond to similar data published in the literature, and the NCCN may have updated these recommendations in anticipation of upcoming FDA approvals. However, this finding further reinforces our previous finding that the NCCN tends to incorporate new drugs quickly, even faster than approval, in contrast to delays in removals.
This study’s implications:
Delays in updating withdrawn indications may lead to doctors prescribing cancer drugs that lack proven survival or quality of life benefits, while exposing patients to the toxicities of those very drugs.
Given that the NCCN recommendations affect insurance coverage, continuing to recommend ineffective drugs may drive unnecessary spending of patients’ and taxpayers’ dollars.
Many cancer drugs are approved based on limited evidence, especially through the accelerated approval pathway. If guidelines rapidly adopt these therapies but hesitate to remove them when confirmatory studies show these drugs are not effective, the result may be persistent low-value care.
Ultimately, this study highlights that the NCCN has competing priorities that are very difficult to balance. The NCCN aims to equip clinicians with as many treatment options as possible that insurers cover and to provide evidence-based recommendations to aid cancer patients. These competing priorities likely contribute to our study’s findings.
Potential Solutions
Have two separate bodies to perform the role of the NCCN. One that provides clinical guidance and another that provides coverage decisions and cost-effectiveness analyses.
Alternatively, a federal standard or law could be established that compendia incorporate and align with FDA regulatory guidance, especially regarding withdrawals.
Many thanks to my mentors, Alyson Haslam and Vinay Prasad, for their guidance, support, and efforts in moving this project forward.
I’ll conclude with the take-home point: guideline makers should continue to quickly adopt new drugs to maximize patient access and inform clinicians, but they should also remove withdrawn drugs with equal efficiency when confirmatory studies call their efficacy into question, leading to FDA withdrawal.





good work... I guess not surprised the withdrawals are slower (sigh)
to the casual observer, seems redundant to have two layers of approvals... if the FDA is assessing efficacy for a drug, then I would think it should be covered for that indication... and vice versa.