World-first tumour-agnostic reimbursement for dual immunotherapy (ipilimumab plus nivolumab), an academic debate, and the DART trial
In two back-to-back essays, we and our counterparts reach opposing conclusions regarding the DART trial and broad reimbursement of dual immunotherapy.
Imagine you have pain. You would probably start with a single painkiller rather than immediately buying and taking a combination of different painkillers. Combining drugs may increase side effects. Besides, why spend more money if one medicine alone might work just as well? The same principle applies in healthcare systems. For the authorization and reimbursement of combination therapies, it is important to show that the combination truly works better than each drug alone. Besides, regulators, payers, and most clinicians will want clinical evidence and, ideally, predictive markers to figure out who’s most likely to benefit. This has been the subject of a vigorous debate between us and our counterparts in the latest issue of the European Journal of Cancer.
To set the stage, in 2025, an unprecedented decision was made by the Australian Pharmaceutical Benefits Advisory Committee (PBAC) to recommend tumour-agnostic reimbursement of nivolumab with or without ipilimumab for all immunotherapy-sensitive advanced or metastatic cancers (see here). The recommendation allows treatment decisions to rely on clinicians’ “appropriate discretion” and “the best available evidence” without providing a clearer definition.
This recommendation could allow for a broad utilization of dual checkpoint inhibition (nivolumab plus ipilimumab) without any biomarker requirement.
Together with our colleague David Thomas, we explain why such a decision sets a concerning precedent. In the same issue of the journal, Vivek Subbiah and Razelle Kurzrock published a counterpoint article as part of a vigorous yet respectful academic debate.
A survival benefit of dual immunotherapy alone over anti-PD(L)1 alone has never been proven
Let’s start with the most immunosensitive tumor type: melanoma. Even in the CheckMate 067 trial, upfront “ipi-nivo” has not been formally shown to be superior to nivolumab alone in terms of overall survival. This is explained here, and summarized in the slide below.
In other tumor types, excluding melanoma, 2 meta-analyses have shown no overall survival benefit of the combination over nivolumab alone (Serritella and Shenoy, Rangwala et al.).
To be clear, dual immunotherapy may still represent an effective strategy in selected settings, for instance in patients progressing after first-line anti-PD(L)1 monotherapy in melanoma. In other tumour types, dual checkpoint inhibition has also demonstrated benefit when compared with control arms other than anti-PD(L)1 monotherapy, such as tyrosine kinase inhibitors in renal-cell carcinoma.
However, in patients with no prior exposure to immunotherapy, the evidence base to date does not support a survival benefit with dual immunotherapy alone over anti-PD(L)1 monotherapy alone in any tumour type.
The DART trial only reinforces our concerns
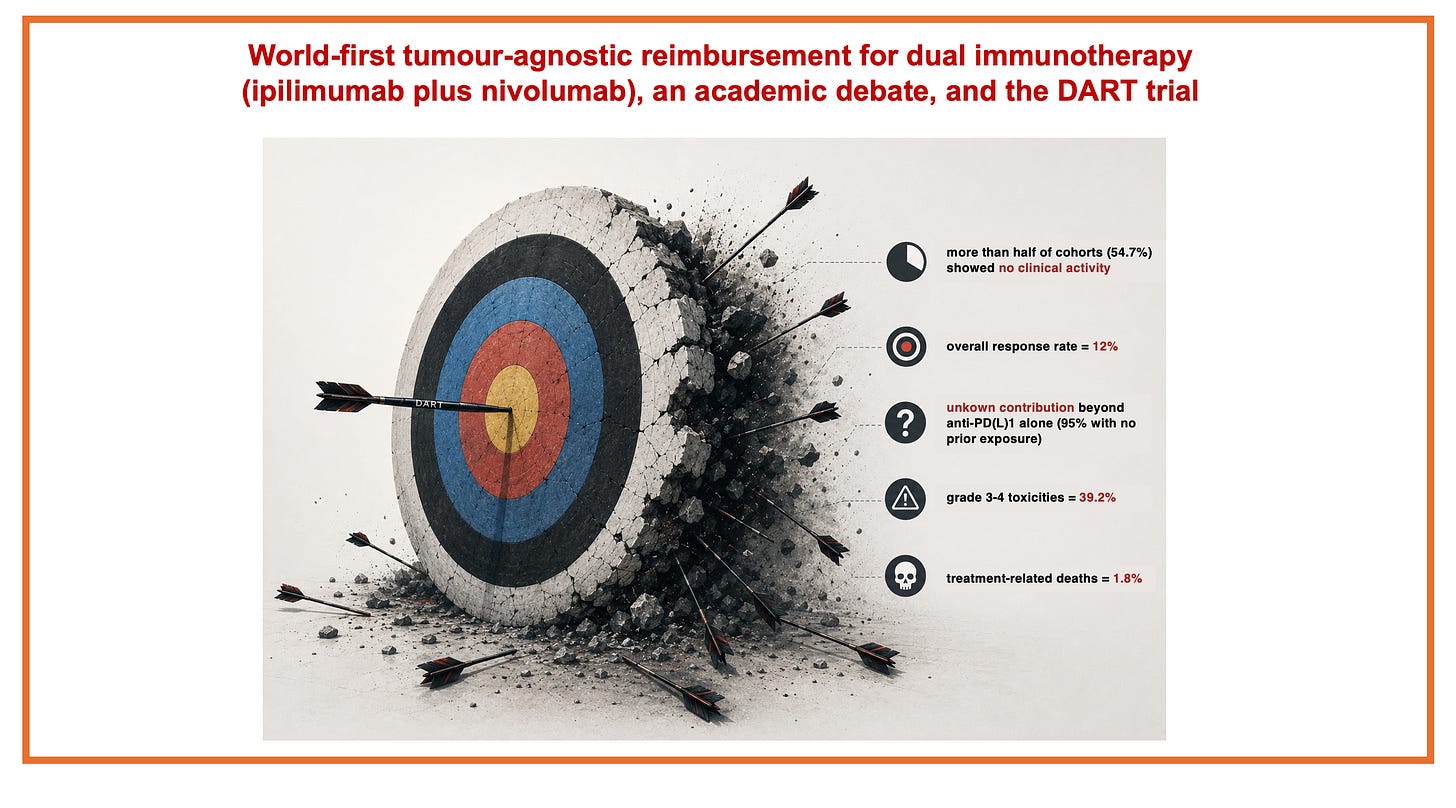
The DART/SWOG S1609 trial was a large phase 2 trial testing ipilimumab (lower dose of 1 mg/kg every 6 weeks) plus nivolumab across many rare cancers. Overall response rate (ORR) was the primary endpoint. The trial took 6 years to enroll 727 patients into multiple rare tumour cohorts. The signal was heterogeneous: more than half of the cohorts had no clinical activity as defined in the trial, while other rare tumour types showed meaningful activity. Across all patients, the median ORR was only 12 %.
While dual checkpoint inhibition had significant activity in some groups, the specific contribution of ipilimumab beyond nivolumab in DART remains unknown: the vast majority of patients (95%) had not received prior anti-PD-(L)1 therapy.
Physical toxicity
A key concern with dual immunotherapy is toxicity.
Immune-related adverse events are very different from those occurring with chemotherapy or oral inhibitors. They can affect any organ, and when high-grade, often requires weeks of corticosteroids. Immune-related toxicity can induce long-lasting impairment, with a potential impact on quality of life.
Again in melanoma, the 10-year follow-up data from the CheckMate 067 trial showed a 63% grade 3-4 rate in patients receiving ipi-nivo, as compared with 25% with nivolumab alone.
The dose used in CheckMate 067 was 3 mg/kg every 3 weeks for 4 cycles. A lower dose, 1 mg/kg, is known to be less toxic, however, still significantly higher as compared with anti-PD(L)1 monotherapy.
In DART, using the low dose of ipilimumab (1 mg/kg every 6 weeks), grade 3–4 treatment-related adverse events were reported in 39.2% of patients. Moreover, treatment-related deaths occurred in 1.8% of patients.
Here is a summary of the DART/ SWOG S1609 trial results:
prospective trials are feasible in rare cancers, and this should be commended
more than half of cohorts (54.7%) showed no clinical activity
overall response rate = 12%
unknown contribution beyond anti-PD(L)1 alone (with 95% of patients with no prior exposure)
grade 3-4 toxicities = 39.2%
treatment-related deaths = 1.8%
As such, we reach different conclusions and disagree that the DART trial supports a broad reimbursement for dual immunotherapy. Rather, we believe it reinforces our concerns.
Financial impact of the reimbursement decision
We noted that in Australia: “The projected expenditure associated with this decision is substantial, exceeding AU$1 billion, with an estimated AU$100–200 million attributable to use beyond existing indications. Funding commitments of this magnitude inevitably carry opportunity costs within a fixed health‑care budget, as resources allocated to one intervention are unavailable for alternative health programmes that may deliver greater population‑level benefit.”
One way forward: a biomarker-driven strategy together with informed data collection
In the Netherlands, the DRUP trial is an adaptive platform trial that provides off-label access to approved targeted therapies and immunotherapies for patients with advanced cancers lacking standard treatment options, while also generating evidence.
Last month, results were published reporting on 1,610 patients within DRUP, who were treated with 37 different off-label targeted or immunotherapies matched to their molecular profile. The report shows that while clinical benefit was seen in some biomarker-defined subgroups, the overall objective response rate (ORR) was only 16%.
The DRUP investigators recommended that: “To maximize patient benefit, […] off-label precision medicines should be used only within frameworks that systematically evaluate efficacy and toxicity, support biomarker refinement and enable stepwise assessment toward potential future label expansion.”
If anything, the conclusion of the DART trial, with a lower ORR than DRUP, should also be that such off-label use requires systematic outcome collection.
It is therefore important that reimbursement decisions that are not backed by clinical evidence, but are instead primarily motivated by addressing unmet need, are accompanied by structured data collection and planned re-assessments. Otherwise, as a health system, you can never know whether you are helping or harming patients. On one hand, you may expose your target population to unnecessary toxicities, and on the other hand, divert healthcare resources away from other patients. In this case, the use of a combination therapy is only justified if monotherapy is not sufficiently effective and this requires use in a trial setting and structured data generation.
Check out our full manuscript for more insights! (openly available here)