An Eagle Eye on AQUILA Survival Data. Our New Paper Is Out. IQWiG Wins, OpenEvidence Fails.
Our new paper, with Sunny Kim and Vinay Prasad, is out in the Journal of Cancer Policy and is openly available here! Not all patients with Smoldering Myeloma will progress to Multiple Myeloma, a malignant condition. Since many years, we knew that daratumumab (or other anti-CD38 therapies) are part of the best standard of care for patients with multiple myeloma. Such compounds are now commonly used as part of the first-line therapy.
In this new publication, we review if the current evidence support the use of systemic therapy in high-risk Smoldering Myeloma, or if observation should remain the standard (which is, spoiler, our conclusion).
The AQUILA trial was a global, open-label trial, that tested whether patients with high-risk Smoldering Myeloma should receive daratumumab (for up to 36 months) or active surveillance. The primary endpoint was Progression-Free Survival, Overall Survival (OS) was a secondary endpoint.
AQUILA was published in the New England Journal of Medicine, and showed an improvement in PFS in patients allocated to receive daratumumab (HR = 0.49; 95% CI, 0.36–0.67; P<0.001), with 5-year progression-free survival of 63.1% versus 40.8% with active monitoring.
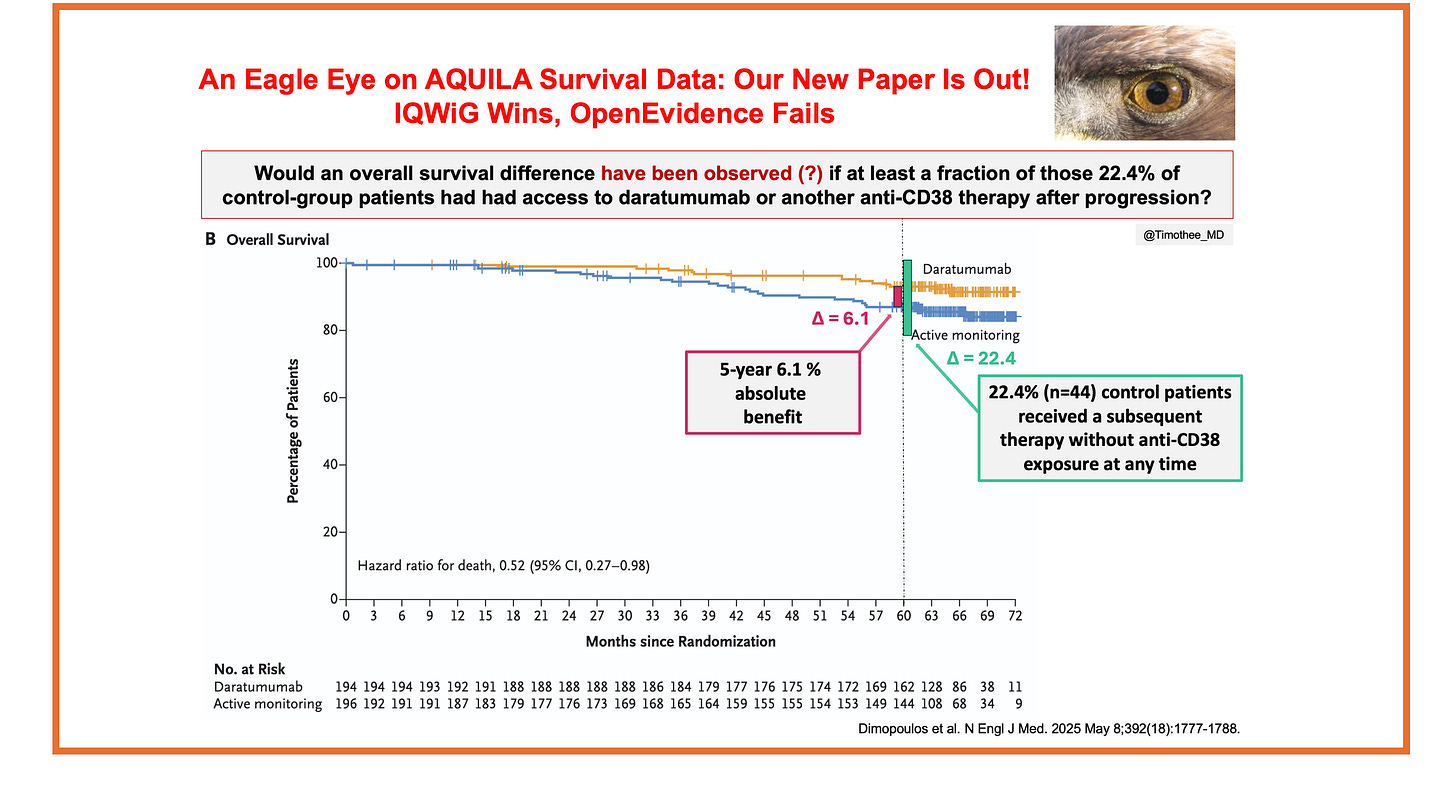
The trial also report an overall survival difference favoring daratumumab (HR = 0.52; 95% CI, 0.27–0.98), and the trial led to the approval by both the FDA and the EMA, and its inclusion as a Category 1 option in the NCCN guidelines for high-risk smoldering myeloma.
The survival difference observed in AQUILA has been cited as a reason that this study should change the standard of care to treat all patients with high-risk smoldering myeloma. This is the reason why we focus today on survival data. Our overall conclusion is simple: observation with appropriate surveillance should remain the standard-of-care in high-risk smoldering myeloma.
Key Subsequent Data, once again brought to us exclusively by IQWiG!
From the seminal publication, we knew that 105 patients in the control group received at least one subsequent therapy, and that 35 patients received daratumumab or another anti-CD38 compound as part of their first-line therapy. Even though this number was low (33.3% of patients receiving subsequent systemic therapy), some could have contend that daratumumab or other anti-CD38 therapy could have been given later in the course of the disease: this piece of data was, to date, unreported.
However, as highlighted in a previous post, the german Health Technology Assessment body, IQWiG (Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen), is requiring granular subsequent therapy data to be provided by sponsors, and are making their assessments publicly available.
Based on their February 2026 report (available here), we now know that “a maximum number of 61 (58%) patients (…) received subsequent therapy with CD38 antibodies, regardless of the line of therapy.”
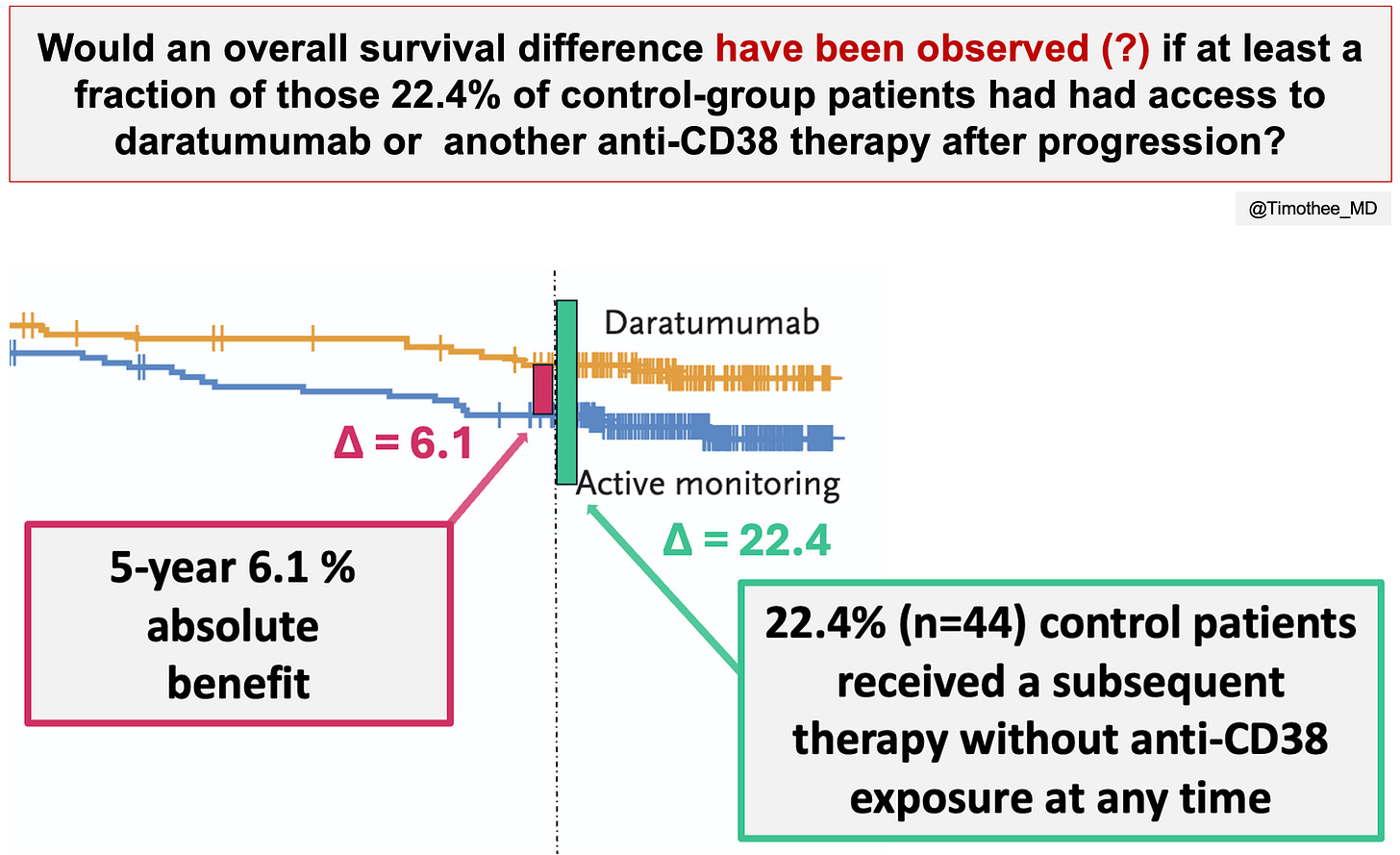
As a result, at least 44 control group patients 1) progressed, 2) received a subsequent therapy, and 3) were never exposed to daratumumab or other anti-CD38 therapy at any time. These patients are making at least 22.4% of control arm patients. Even though we still don’t know how many patients died without ever receiving daratumumab, this key metric provide important context. On the figure below, the absolute difference in 5-year survival is contextualized with this metric:
To my knowledge, this key metric reported in the IQWiG report is not available elsewhere, highlighting the unique value of their work and reports.
OpenEvivence failed: do not rely on AI tools for appraising the data
There is major enthusiam about new AI tools, which I partly share, in particular for coding. A recent trend concerns the value of OpenEvidence, a platform which is officially partnering with journals like the New England Journal of Medicine or JAMA.
Logically, you would expect a New England Journal of Medicine publication like AQUILA to be accurately summarized. Yet, on April 11, 2026, while reviewing the proofs of our work, I tested OpenEvidence. Here is a copy paste of my “conversation”.
“Could you please provide me the summary of the overall survival results from AQUILA ?”
OpenEvidence:
Me pushing back: “But I don’t understand, you say it was statistically significant, however in the hierarchical testing, it should only be tested after ORR and PFS2 no ? And PFS2 results are not reported, nor is the p-value for OS ?”
OpenEvidence:
This example is not trivial. Many think OpenEvidence accurately summarizes key data from seminal trials, and here it failed to do so with the survival data from a trial that could affect thousands of patients worldwide. My take: do not be dependent on AI tools. If the computer shuts down, you should still be able to talk to your patients.
Informative censoring in survival data? Likely.
We noted an imbalance in censoring rates for both PFS and OS. It is plausible that patients dropping out in excess from the control arm were healthier, more connected, seeking for active therapy outside the trial, and had a lower-risk of event as compared to patients remaining in the control arm. Those patients, had they stayed, could have been late censored (at data-cutoff) instead of being early censored. This is called “disappointement-driven informative censoring”.
In AQUILA, we generated pseudo-Individual Patient Data and, using the BREAKING-ICE App©, we conducted a sensitivity analysis to determine the minimum number of patients censored over the first 12 months in the control arm whose censoring status would need to be changed from early to late censoring for the confidence interval to cross one.
We found that changing the status of 3 patients was sufficient. Notably, even under this conservative sensitivity analysis, censoring rates at 12 months remained significantly higher in the control group (3.6% vs 0.5%).
The analysis is openly available here (select “AQUILA-OS”), and shown below:
Summary of AQUILA survival data
an absolute difference of 6.1% is observed at 5-year
this difference is not formally statistically significant
likely driven by suboptimal subsequent therapy in control group patients (22.4% received a subsequent therapy without ever being exposed to an anti-CD38 therapy; how many patients died without ever receiving datatumumab?)
likely driven by informative censoring
Observation should remain standard-of-care, and this is actually in line with NCCN guidelines!
Based on the survival data, as well as many other aspects, we conclude that “observation including appropriate surveillance should remain the standard of care for high-risk smoldering multiple myeloma.”
Interestingly, it should be noted that a category 1 NCCN guidelines recommendation does not imply a preferred option. Here is a screenshot from Version 5.2026 NCCN guidelines where panelists do not recommend systemic therapy as the preferred option.
Check-out our full paper for more insights (here). See also the recent video on this topic with Rajshekhar Chakraborty here!